Nifty rebounds
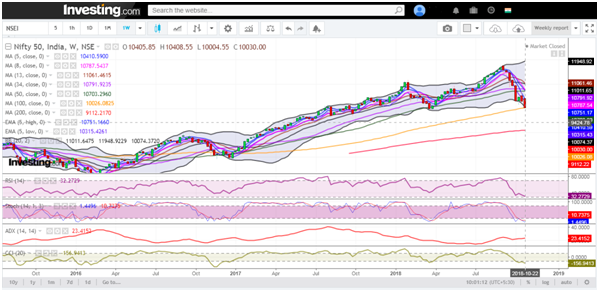
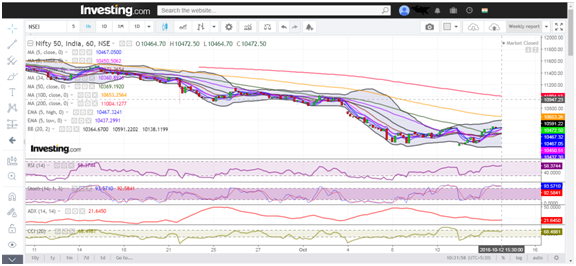
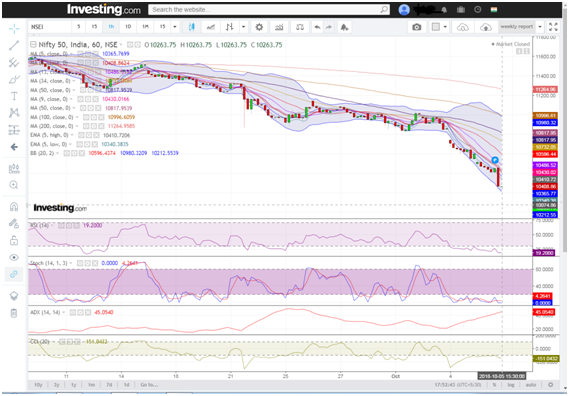
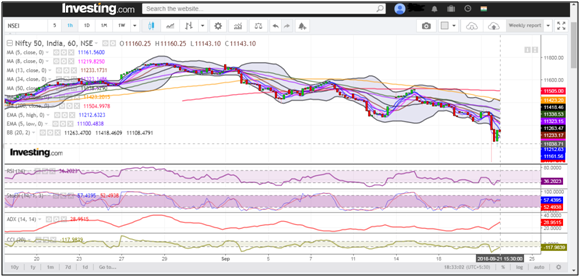
Benchmark Nifty 50 had a positive close this week, rallied over 170 points to trade back to levels seen 3 weeks back @ 8000. With Friday’s 68 points gap up, the index finally rose above the crucial resistance of 7900. Looking at the weekly charts, it is clear that 8200 is the immediate resistance to watch for. On the downside, we sense that rallies will face resistances in the range of 8200.
Sectors end in Green
With the dramatic rise in the national index, majority of the sectoral indices were outperforming.
- CNX AUTO which has its immediate support at 7500 and resistance at 8200 lost 0.19% this week to close at 7842.
- CNX ENERGY lost gained 4% this week to close at 7805. It took its major support at 7400 gaining ever since.
- CNX FINANCE too gained the same way with the rebound at the support of 6600, the sector ended the week with a positive change of 4%.
- CNX FMCG had a flat week compared to its counterparts. With the gain about 0.44%. Its immediate support is at 18768 and the resistance is at 20240.
- CNX IT gained this week with a rise of 2.58%. Currently trading at 11726, its immediate support is at 10990 while the resistance is at 12323.
- CXN MEDIA gained this week with a rise of 1.33%. Currently trading at 2372, its immediate support is at 2140 while the resistance is at 2500.
- CNX METAL seems to be nearing a reversal. Currently trading at 1790, its immediate support is at 1700 while the resistance is at 2145.
- CNX PHARMA gained this week with a rise of 3.32%. Currently trading at 12751, its immediate support is at 11867 while the resistance is at 13785.
- CNX PSU BANK gained this week with a rise of 5.11%. Currently trading at 3147, its immediate support is at 2690 while the resistance is at 3730.
- CNX REALTY gained this week with a rise of 3.51%. Currently trading at 171, its immediate support is at 162 while the resistance is at 180.
August wholesale inflation contracts sharply to -4.95% vis-a-vis -4.05% in July– The WPI inflation has contracted sharply for the 10th straight month to -4.95% in the month of August versus -4.05% in July influenced by global deflationary trend in commodity prices. The manufactured index declined to -1.92% against -1.42% in the previous month, but the primary article group rose by 1.6% as its components food articles and non-food articles rose by 2.6% and 0.8%, respectively. The third component — minerals — declined by 4.4%. Another major group fuel and power with a weight of 14.91% in the WPI basket declined by 4.4% as commodity prices collapsed. The declining WPI data may give rise to rate cut in the upcoming RBI monetary policy.
Tata JLR making its entry into SUV market with launch of F-Pace model– Tata JLR has entered the SUV market with the launch of F-Pace model which is expected to hit the Indian market in the second half of 2016. The F-Pace SUV launched at the Frankfurt Motor Show has been dubbed the “ultimate practical sports car”, which embodies the spirit of the iconic British luxury car brand with modern technology. The F-Pace SUV is expected to hit the roads in Europe and the US in a phased rollout by early next year, with an Indian launch expected in the latter half of 2016. By the way you know, It has made its entry at the show with a spectacular ‘Guinness Book of Records’ breaking drive around a 19.1-metre-high loop driven in a daring stunt by racing driver Terry Grant.
Granules India gets USFDA nod for painkiller drug– The company has received an approval from the US Food and Drug Administration (USFDA) for its painkiller drug. The US FDA has approved Granules’ abbreviated new drug application for Ibuprofen tablets USP, 400 mg, 600 mg and 800 mg.
Jubilant Life gets USFDA nod for anti-migraine tablets – The company has received nod from the US Food and Drug Administration (USFDA) for its abbreviated new drug application (ANDA) for zolmitriptan orally disintegrating tablets in strengths of 2.5 mg and 5mg, . The company’s drug is a generic version of AstraZeneca’s Zomig-ZMT tablets, which is used for acute treatment of migraine headaches in adults. As on June 30, Jubilant Life Sciences had a total of 815 filings for formulations of which 389 have been approved in various regions globally. This includes 72 ANDAs filed in the US, of which 38 have been approved and 46 dossier filings in Europe.